Effective treatment of
allergic diseases
such as asthma and atopic dermatitis
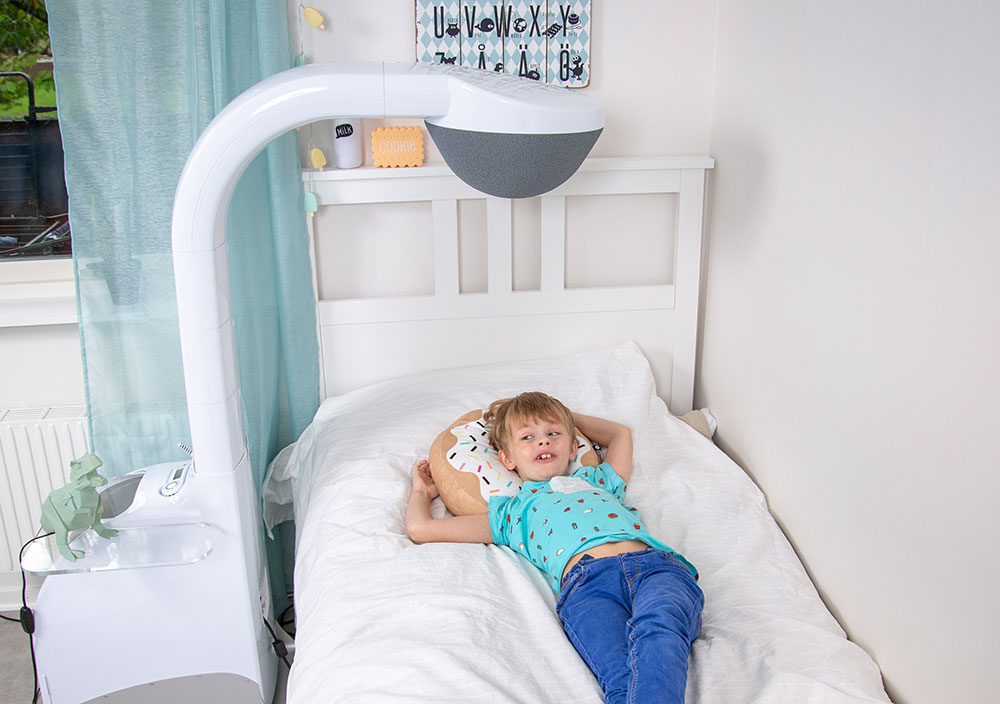
Airsonett® Air4 is a medical device for effective home treatment of allergic asthma and atopic dermatitis. The patented TLA treatment (Temperature-controlled Laminar Airflow) provides 99.5% clean air in the breathing zone. Your airways and immune system can rest and recuperate when you sleep, and you wake up rested, ready for a new day.
Treatment with TLA is recommended by the Swedish National Board of Health and Welfare (Socialstyrelsen) and the Swedish paediatricians ‘allergy association (BLFA). It has also been reviewed in the UK by The National Institute for Health and Care Excellence (NICE) and proven cost effective by Health Improvement Scotland. Airsonett Air4 is a CE-marked medical device that meets the requirements according to MDR 2017/745, Class 1.
Effective
- Reducing exacerbations1,2
- Improved control of allergic diseases1,3,4,5,6
- Improved quality of life3,4,5
- No pharmacological side effects1,2,3,4,5,6
- Cost-effective7,8
1: Schauer et al, 2:Wang et al; 3: Boyle et al, 4: Pedroletti et al, 5: Gore C et al, 6: Traidl et al, 7: Brazier et al, 8: Chauhan et al

Economical
Health economics studies confirm lower treatment costs compared to pharmacological treatment options, fewer clinic visits and better control over patient symptoms.

This is how it works
Airsonett Air4 creates a zone of 99.5% clean air, free of particles and allergens around the face during night sleep. The airways and immune system rest and can withstand allergens even during the day.
Life with Airsonett
Airsonett Air4 helps many patients with allergic asthma and atopic dermatitis, in Sweden and the UK, to better health and a better quality of life.
News
Read news on Airsonett, see press releases and get in touch here.